Page History
...
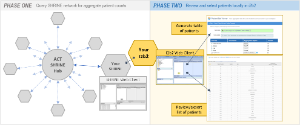
The Accrual to Clinical Trials (ACT) project goal is to create a federated network of National Clinical and Translational Science Award (CTSA) Consortium institutions to significantly increase participant accrual to the nation’s highest priority clinical trials. PHASE ONE consisted of the formation of a SHRINE network of participating institutions in order to query for aggregate patient counts across all sites in real-time. PHASE TWO continues the workflow at the local site by enabling the reviewing and selection of patients locally in i2b2.
How-to Guides:
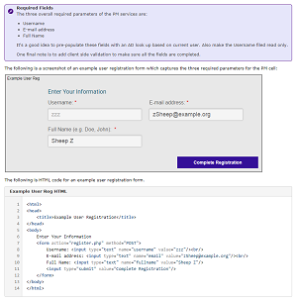
Accrual to Clinical Trials i2b2 Community Wiki How-to: Create a User Registration AppIn this guide, we demonstrate how you can create a user registration form that will allow users to self register for SHRINE access. The services offered by the PM Cell make online user registration possible.
| |||||||||
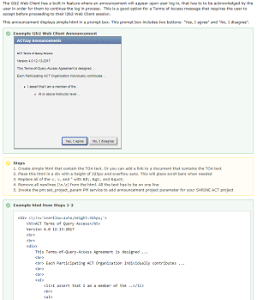
| Accrual to Clinical Trials i2b2 Community Wiki | How-to: Automatic Terms of AccessThe SHRINE web client have a built in feature where an announcement can appear upon user log in. This guide will show how this acknowledgement dialog can be used for the Terms of Access for the ACT project.
|
Software:
Accrual to Clinical Trials i2b2 Community WikiIn this guide, we demonstrate how you can create a user registration form that will allow users to self register themselves for SHRINE access. The services offered by the PM Cell makes online user registration possible | Phase Two SoftwareRead about the ACT Phase Two Software.
|